Ascophyllan HS is a standardized hot water extract from Ascophyllum nodosum, the large brown seaweed harvested off the coasts of Ireland, Norway and Canada. The active compound - ascophyllan - is a sulfated polysaccharide structurally distinct from fucoidan, with significantly higher uronic acid content driving stronger immune-modulating activity. Manufactured in Japan by a specialist seaweed processing partner, the extract is standardized to a minimum of 20% ascophyllan and processed with activated carbon to deliver a clean profile: low heavy metals, minimal viscosity, neutral taste and reduced seaweed odor. The ingredient is patent-protected for infectious pneumonia prevention (Japan Patent No. 6854613) and validated through human clinical trials, animal studies and cellular research published in peer-reviewed journals including International Journal of Biological Macromolecules, Marine Drugs and Anticancer Research. With a single 100 mg daily dose, Ascophyllan HS delivers measurable benefits across three commercial verticals: immune support, metabolic health (GLP-1 + alpha-glucosidase) and oral care.
Both ascophyllan and fucoidan are sulfated polysaccharides from brown seaweeds, but their structures are fundamentally different - and that difference drives bioactivity. Fucoidan is built around fucose with sulfate groups. Ascophyllan is built around uronic acid, with xylose and fucose as side chains. The numbers are stark: ascophyllan contains 21.4% uronic acid versus 5.8% in fucoidan (3.7x more), and 13.4% xylose versus 4.3% (3.1x more). This structural distinction translates into measurable functional differences. In comparative in vitro studies, ascophyllan induced stronger cytokine production (IL-6, IL-12, TNF-alpha) from dendritic cells than fucoidan and triggered higher nitric oxide secretion from macrophages. NK cell cytotoxicity against tumor cells was activated more efficiently. The dose required for measurable immune effect drops from 300 mg/day for typical fucoidan supplements to 100 mg/day for Ascophyllan HS.

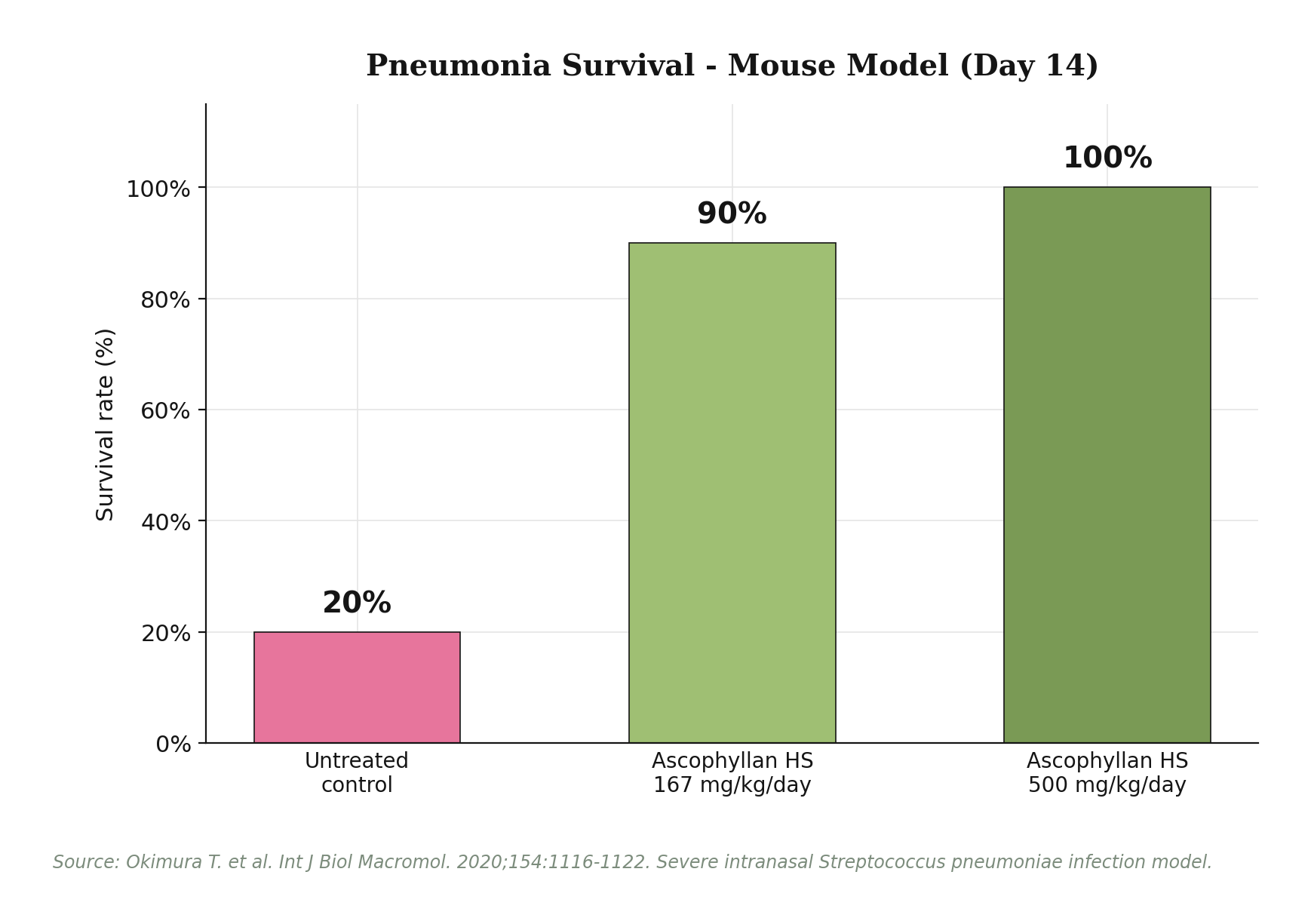
Immune support: NK cells, dendritic cells, cytokines. A human clinical trial in adults aged 40-59 with low baseline NK activity showed that 8 weeks of Ascophyllan HS at 100 mg/day significantly increased NK cell activity compared with placebo (p<0.05) and elevated serum interferon-gamma (IFN-gamma) and interleukin-12 (IL-12). Animal research extended these findings with a striking result: in a severe intranasal Streptococcus pneumoniae infection model, untreated control mice had 80% mortality by day 14, while mice receiving oral Ascophyllan HS at 167 mg/kg/day showed 90% survival, and the 500 mg/kg/day group reached 100% survival. Histopathology confirmed prevention of typical pneumonia features - inflammatory infiltration, edema, fibrin deposition. This patented anti-pneumonia mechanism (Japan Patent No. 6854613) operates through activation of host immunity rather than direct antibacterial action.
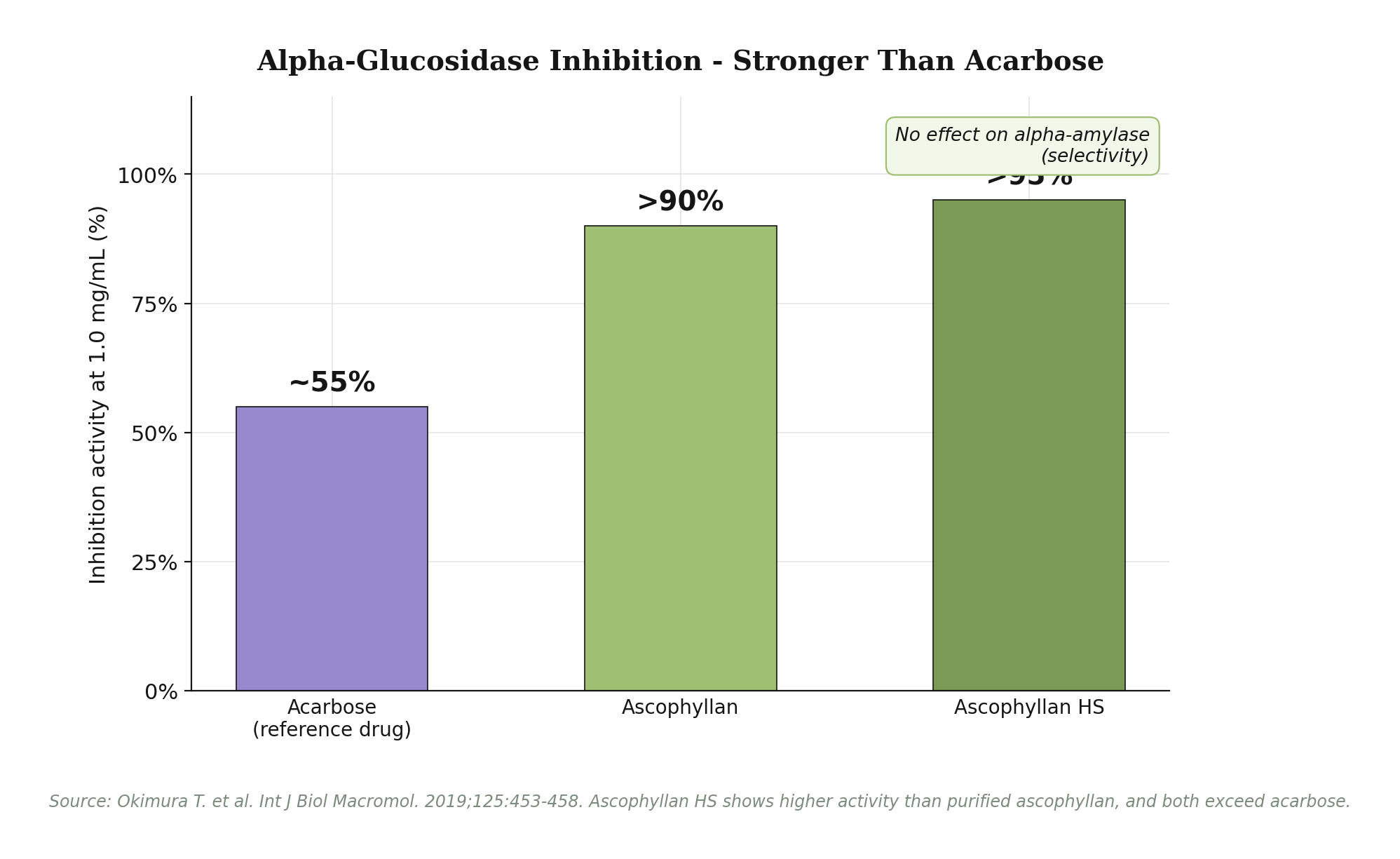
Metabolic health: dual mechanism via GLP-1 and alpha-glucosidase. Ascophyllan HS targets postprandial glucose through two complementary pathways. First, it stimulates GLP-1 (glucagon-like peptide-1) secretion from human intestinal NCI-H716 L-cells in a dose-dependent manner. GLP-1 is the same incretin hormone targeted by injectable Ozempic and Saxenda - it suppresses appetite, slows gastric emptying and stimulates glucose-dependent insulin secretion. Second, ascophyllan inhibits alpha-glucosidase, the enzyme responsible for carbohydrate breakdown. Inhibition exceeded 90% at 1.0 mg/mL, with Ascophyllan HS showing higher activity than purified ascophyllan and stronger activity than acarbose, the reference antidiabetic drug. Critically, ascophyllan does not inhibit alpha-amylase, providing selectivity that minimizes the gastrointestinal side effects associated with non-selective inhibitors. In an 8-week human clinical trial at 100 mg/day, oral glucose tolerance test AUC values were significantly lower than placebo, and HbA1c showed a downward trend in the treatment group only.

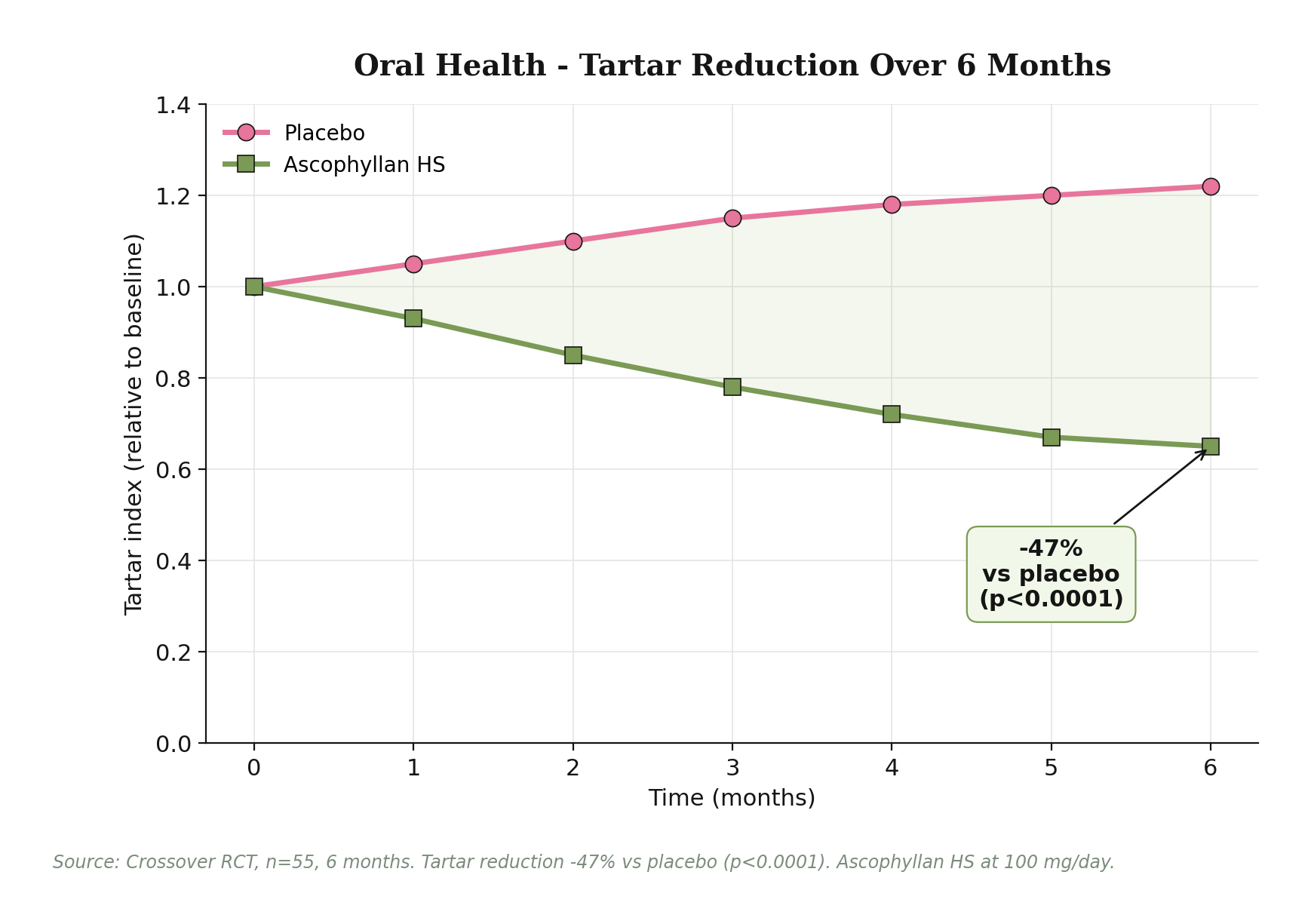
Oral and pet care: tartar, plaque, halitosis. A 6-month crossover study (n=55) with 100 mg/day Ascophyllan HS demonstrated tartar reduction of 47% versus placebo (p<0.0001), along with reduction of dental plaque (Quigley-Hein index, p=0.008). The mechanism extends to volatile sulfur compound (VSC) suppression - the chemicals responsible for halitosis. A canine study with 60 dogs receiving Ascophyllum nodosum supplement at approximately 210 mg/day showed significant VSC reduction from day 60 onwards. This positions Ascophyllan HS as a dual-channel ingredient for both human oral care supplements and the growing pet oral care category.
Appearance and texture. Brown free-flowing powder with smooth, uniform particle size optimized for consistent dispersion across tablet, capsule, sachet and beverage applications.
Taste and odor. Activated carbon treatment during manufacturing significantly reduces the marine odor characteristic of raw seaweed extracts. At the recommended 100 mg per serving, residual taste impact is minimal and easily masked.
Solubility and viscosity. Excellent water solubility at concentrations from 0.1% to 2.0%. Critically, the alginate-class viscosity-building polysaccharides have been removed during processing, meaning Ascophyllan HS does not thicken liquid formulations. This is a meaningful advantage for clear functional beverages and ready-to-drink products where competing seaweed extracts create undesirable mouthfeel.
The validated daily intake is 100 mg of Ascophyllan HS, supported by human clinical trials for immune support, glucose management and oral health, and patent claims for pneumonia prevention. Continuous intake over 8 weeks or longer is recommended to achieve measurable immune and metabolic effects. The 100 mg dose can be delivered as a single serving or split (50 mg twice daily); both produce equivalent results in published protocols. For metabolic targets, intake before or with meals capitalizes on alpha-glucosidase inhibition during carbohydrate digestion.
Capsules and tablets. The dominant format. Low bulk density and good flowability make Ascophyllan HS suitable for high-speed automated capsule filling and tablet compression. Frequent stack partners include vitamin C, vitamin D3, zinc, beta-glucans (immune); berberine, chromium picolinate, mulberry extract (metabolic); CoQ10 and probiotics (oral health).
Functional beverages and powder mixes. The low-viscosity, neutral-taste profile makes Ascophyllan HS one of the few seaweed polysaccharides usable in clear ready-to-drink wellness shots and reconstitutable powder blends without altering mouthfeel. Compatible with pasteurization.
Jelly sticks and gummies. Established Japanese format, particularly for immune and metabolic positioning. Disperses well into gel matrices without affecting set texture. The brown color suggests pairing with coffee, chocolate, hojicha or maple flavor profiles for gummy lines.
Pet supplements. Multiple commercial canine and feline oral-care products already incorporate Ascophyllum nodosum-based ingredients in the Japanese and US markets. Ascophyllan HS offers a standardized, patent-supported version for premium pet brands.
Ascophyllan is a sulfated polysaccharide classified as a xylofucoglycuronan, isolated from the brown seaweed Ascophyllum nodosum. Molecular weight ranges from 100,000 to 300,000 Daltons, placing it firmly in the high-molecular-weight water-soluble fiber category. The structural backbone is uronic acid, with xylose and fucose attached as side chains and sulfate groups distributed across the molecule. This composition places ascophyllan in a structurally distinct class from fucoidan despite their shared brown seaweed origin and shared sulfate functionality. The structural difference is biologically meaningful - ascophyllan and fucoidan engage immune receptors and metabolic targets with measurably different efficiency.
Ascophyllum nodosum is a large brown alga - reaching up to 2 meters in length - that grows along the cold North Atlantic coastlines of Ireland, Norway, Iceland and Canada. It does not occur naturally in the Pacific Ocean or along Japanese shores. In Northern European coastal communities the seaweed has centuries of use as food (consumed raw in salads, brewed as tea), as livestock fodder, and as an organic mineral supplement for soil enrichment. Its nutritional content - amino acids, polysaccharides, trace minerals - was understood empirically long before its bioactive components were chemically characterized. Scientific isolation and structural identification of ascophyllan as a distinct compound was led by Japanese researchers, with foundational work by Tatsuya Oda and colleagues at Nagasaki University in collaboration with a Japanese industrial partner. Sustained research from approximately 2009 onwards established ascophyllan's distinct profile in immune modulation, glucose regulation and oral health.
Ascophyllan engages the immune system through a coordinated sequence. At the entry point, it triggers maturation of dendritic cells (DCs), upregulating co-stimulatory molecules CD40, CD80, CD86 and MHC class I and II expression. Mature DCs release IL-6, IL-12 and TNF-alpha, which in turn drive Th1 (T helper type 1) and Tc1 (cytotoxic T cell type 1) responses - the immune pathways responsible for defense against intracellular pathogens (viruses, bacteria) and tumor surveillance. NK cell activation occurs both directly and through DC-mediated cytokine signaling. In mouse models, ascophyllan increased the population of IFN-gamma-producing NK cells in the spleen and elevated cytotoxicity against Yac-1 target cells. Comparison with fucoidan in identical experimental conditions consistently showed ascophyllan as the more potent activator. The downstream consequence - validated in the pneumococcal pneumonia model - is enhanced host defense without the inflammation overshoot that limits some immune-stimulating ingredients.
The metabolic mechanism operates through two parallel pathways converging on postprandial glucose control. The first is GLP-1 secretagogue activity. Ascophyllan stimulates intestinal L-cells (NCI-H716 line) to release GLP-1 in a dose-dependent fashion at concentrations from 1 to 100 ng/mL. GLP-1 acts on the hypothalamus to suppress appetite, slows gastric emptying to extend satiety, and amplifies glucose-dependent insulin release from pancreatic beta cells. The second pathway is direct enzymatic inhibition. Ascophyllan binds alpha-glucosidase, the brush-border enzyme that breaks dietary carbohydrates into absorbable monosaccharides, reducing its activity by more than 90% at 1.0 mg/mL. The Ascophyllan HS commercial preparation shows even higher inhibitory activity than purified ascophyllan, and both significantly outperform acarbose - the reference oral antidiabetic drug. Notably, ascophyllan does not inhibit alpha-amylase, the upstream salivary and pancreatic enzyme. This selectivity matters: non-selective inhibitors of both alpha-glucosidase and alpha-amylase frequently cause flatulence, bloating and diarrhea due to undigested carbohydrate fermentation in the colon.
The Japanese patent holder owns Japan Patent No. 6854613, covering compositions for the prevention and treatment of infectious pneumonia using ascophyllan. The patent is supported by the Okimura et al. 2020 mouse infection model published in International Journal of Biological Macromolecules. Use of the patent claims on finished products requires prior coordination with the patent holder and meeting the minimum 100 mg/day dose threshold. Iizuka Shoukai handles this coordination on behalf of B2B clients formulating with the ingredient.
Ascophyllan HS is standardized to a minimum 20% ascophyllan content, quantified by the method published in Nakayasu et al. 2009 (Biosci Biotechnol Biochem). Manufacturing combines hot water extraction with a sequence of purification steps engineered to eliminate the limitations of crude seaweed extracts. Citric acid pretreatment, centrifugal separation and activated carbon treatment remove heavy metals, alginate viscosity polymers and the volatile compounds responsible for marine odor. The result: lead under 20 ppm, arsenic under 10 ppm, total aerobic count under 3,000 CFU/g, coliform negative. The 400-pesticide screen and 40-veterinary-drug screen return non-detect across all targets. Ames mutagenicity test is negative. Acute oral toxicity LD50 in mice exceeds 2,000 mg/kg - twenty times the recommended human daily dose normalized for body weight.
Immune support formulas targeting respiratory health, particularly in the 40+ demographic where natural NK activity declines. Suitable as standalone or in combination with vitamin D3, zinc and elderberry extract.
Metabolic and weight management products riding the GLP-1 category expansion. Positioned as natural complement or alternative to glucose modulators. Effective with or without other glycemic agents (chromium, berberine, mulberry).
Oral care supplements and toothpastes targeting tartar prevention, plaque reduction and breath freshness. Strong fit for the periodontal health and senior oral care segments.
Pet oral care for dogs and cats. The Japanese pet supplement market has multiple established Ascophyllum-based products; Ascophyllan HS offers a standardized, patent-backed upgrade.
Functional foods and beverages beyond traditional supplements - immune shots, wellness jelly sticks, fortified teas and breath-freshening confectionery.
【1】 Okimura T, Jiang Z, Komatsubara H, Hirasaka K, Oda T. Therapeutic effects of an orally administered edible seaweed-derived polysaccharide preparation, ascophyllan HS, on a Streptococcus pneumoniae infection mouse model. Int J Biol Macromol. 2020;154:1116-1122.
【2】 Okimura T, Jiang Z, Liang Y, Yamaguchi K, Oda T. Suppressive effect of ascophyllan HS on postprandial blood sugar level through the inhibition of alpha-glucosidase and stimulation of glucagon-like peptide-1 (GLP-1) secretion. Int J Biol Macromol. 2019;125:453-458.
【3】 Zhang W, Du JY, Jiang Z, Okimura T, Oda T, Yu Q, Jin JO. Ascophyllan purified from Ascophyllum nodosum induces Th1 and Tc1 immune responses by promoting dendritic cell maturation. Mar Drugs. 2014;12(7):4148-4164.
【4】 Nakayasu S, Soegima R, Yamaguchi K, Oda T. Biological activities of fucose-containing polysaccharide ascophyllan isolated from the brown alga Ascophyllum nodosum. Biosci Biotechnol Biochem. 2009;73(4):961-964.
【5】 Jiang Z, Okimura T, Yamaguchi K, Oda T. The potent activity of sulfated polysaccharide, ascophyllan, isolated from Ascophyllum nodosum to induce nitric oxide and cytokine production from mouse macrophage RAW264.7 cells. Nitric Oxide. 2011;25(4):407-415.
【6】 Abu R, Jiang Z, Ueno M, Isaka S, Nakazono S, Okimura T, Cho K, Yamaguchi K, Kim D, Oda T. Anti-metastatic effects of the sulfated polysaccharide ascophyllan isolated from Ascophyllum nodosum on B16 melanoma. Biochem Biophys Res Commun. 2015;458(4):727-732.
【7】 Yoshida T, Bi Y, Jin JO et al. Ascophyllan induces activation of natural killer cells in mice in vivo and in vitro. Mar Drugs. 2019;17(4):197.

Both are sulfated polysaccharides from brown seaweeds, but their molecular skeletons differ. Fucoidan has a fucose backbone (28.4% fucose) with sulfate groups (19.4%). Ascophyllan has a uronic acid backbone (21.4% uronic acid - 3.7 times more than fucoidan) with xylose (13.4% - 3.1 times more) and fucose as side chains. This drives stronger immune activity: in comparative studies, ascophyllan triggered more potent dendritic cell maturation and NK cell activation. The effective daily dose drops from approximately 300 mg for typical fucoidan supplements to 100 mg for Ascophyllan HS - a 3x potency advantage at the formulation level.
Iizuka Shoukai supplies Ascophyllan HS at 500 USD per kilogram with an MOQ of 1 kg. Standard lead time from Japan is 3 to 5 weeks for orders up to 50 kg, with larger volumes negotiable. The product is export-ready, GMP-grade, and supplied with full documentation: Certificate of Analysis, MSDS, allergen declaration, and patent license arrangement. Iizuka Shoukai handles export logistics from Japan to USA, EU, UAE, CIS, Australia and 20+ other markets, including DDP terms on request. Sample sachets are available for formulation trials before commercial purchase.
The clinically validated dose is 100 mg per day, which can be delivered as a single 100 mg serving or split into two 50 mg doses with equivalent results. Ascophyllan HS is a low-viscosity, neutral-tasting brown powder that performs across formats: capsules (single ingredient or in synergistic blends), tablets, sachet sticks, beverages, jellies and gummies. It is heat-stable up to standard pasteurization temperatures and water-soluble. For metabolic positioning, intake with meals capitalizes on the alpha-glucosidase inhibition mechanism. For immune positioning, daily dosing without timing constraints is sufficient.
Most Chinese suppliers offer crude fucoidan or undefined seaweed polysaccharide blends with variable potency, no clinical validation, and no patent protection. Ascophyllan HS is a single-source standardized extract from Ascophyllum nodosum, manufactured in Japan under GMP, with Japan Patent No. 6854613 for infectious pneumonia prevention and four published human, animal and cellular studies. Standardization is guaranteed at minimum 20% ascophyllan content via activated carbon processing. Heavy metal load is minimized through Japanese-controlled inputs and processing. Iizuka Shoukai supplies the export channel with full COA per batch and traceability back to specific harvest seasons.